Methane-Air Reaction Mechanism (GRI-Mech 3.0)
In this sample, the methane-air reaction mechanism from GRI-Mech 3.0 [1] is used. The mechanism consists of 325 reactions that involve 53 species. The three following problems were solved to compare our results to independent calculations of other authors.
NO Emission in Methane Oxidation
Parameters of this problem are similar to that studied in [2]. We don’t concern about the main subject of the work [2] (creating a reduced reaction mechanism for NO emission) and take only the numerical data for NO reburning. A fixed pressure problem is solved.
Input values
Pressure: 1atm
Temperature: 1300K or 1600K
Table 1. Initial mole fractions
CH4 | C2H6 | O2 | NO | H2O | N2 |
2.864E-03 | 2.98E-04 | 5.09E-03 | 9.47E-04 | 2.16E-02 | 0.9692 |
Solution
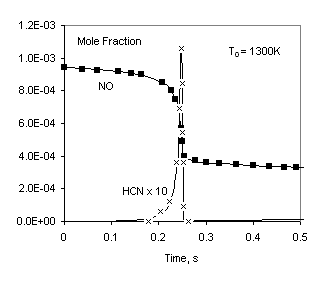
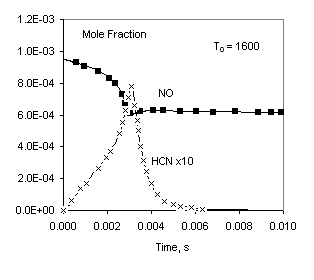
Fig 1. Comparison of calculated profiles: points – data from [2], lines – Chemked calculation.
Thermal decomposition of CH2O at fixed pressure and fixed temperature (from the GRI-Mech collection)
Conditions of this calculation correspond to the experiments [3].
Input values
Temperature: 1805K
Mole fractions: [CH2O]: [AR] = 4 : 96
AR concentration : 1.9E-05 mole/cm3
Solution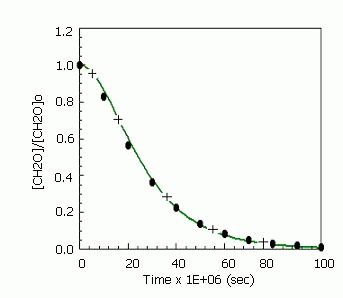
Fig 2. Normalized CH2O profile: circles – experimental data [3], line – calculation [1], crosses – Chemked calculation
Oxidation of methane at fixed pressure and fixed temperature (from the GRI-Mech collection)
Conditions of this calculation correspond to the experiments [4].
Input values
Pressure: 1atm
Temperature: 2454K
Mole fractions: [CH4]: [O2]: [AR] = 0.1 : 0.4 : 99.5
Solution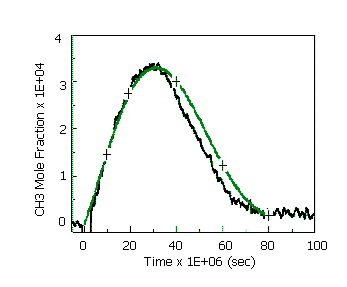
Fig 3. Mole fraction of CH3:
black line – experiments [4], green line – calculation [1], crosses – Chemked calculation
References
[1] GRI-Mech 3.0, The Gas Research Institute,
http://www.me.berkeley.edu/gri_mech/
[2] C.J.Sung, C.K.Law, and J.-Y.Chen. Augmented Reduced Mechanisms for NO Emission in Methane Oxidation. Combustion and Flame, 125, p.906 ( 2001).
[3] Y. Hidaka , T. Taniguchi, H. Tanaka, T. Kamesawa, K. Inami, and H. Kawano. Shock-Tube Study of CH2O Pyrolysis and Oxidation, Combustion and Flame, 92, p. 365 (1993).
[4] A.Y. Chang, D.F. Davidson, M. DiRosa. R.K. Hanson, and C.T. Bowman. Shock Tube Experiments for Development and Validation of Kinetic Models of Hydrocarbon Oxidation, 25th Symposium (International) on Combustion, Poster 3 - 23 (1994).